(UPDATED) MANILA. Philippines — When does a problem become worse? One is if it isn’t solved, and two is if the problem is made serious by a supposed solution that turns out to be another problem.
Residents of Hagonoy, Bulacan are caught in this kind of quandary now and have realized that prevention isn’t always the best cure after French pharmaceutical giant Sanofi Pasteur’s admitted that its product — Dengvaxia — supposedly the world’s only answer to the dengue problem — poses risks among those who had themselves injected with the vaccine prior to contracting the mosquito-borne disease.
And Hagonoy residents have all the reasons to be extra worried: the first class town, they claim, is among the areas in Bulacan most vulnerable to dengue. Thus they fear that many school children, who didn’t have dengue before, might catch the illness that could become severe because they received shots of Dengvaxia.
The Provincial Health Office of Bulacan recorded a total of 7,582 persons — mostly aged one to 10 — who got infected with the dengue virus from January 1 to December 2, 2017 — a 48-percent jump from the number of cases listed last year.
Visitacion Escartin is among Hagonoy residents who worry over the safety of their children.
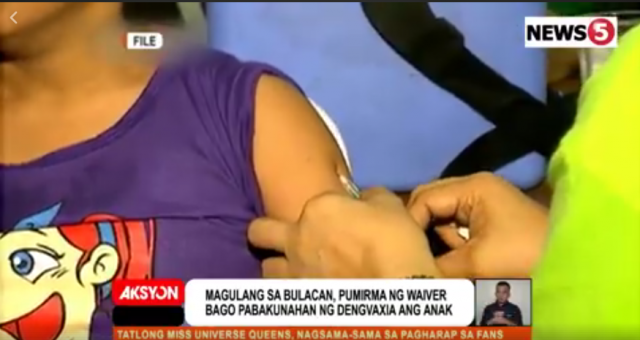
Escartin said that at first, she was thankful that she didn’t have to buy the expensive vaccine to protect her child from dengue.
When the school announced last year that it would be giving her child and other students complete Dengvaxia shots, Escartin did not think twice. She signed a consent form allowing the school to have her then nine-year-old child injected with the vaccine thrice every six months.
“S’yempre po, iga-grab ko agad kasi libre. Tapos, magiging safe pa ang anak ko sa dengue. Kasi that time po, usong-uso ang dengue, kaya hindi na ako nagdalawang isip, pumirma na ako sa waiver,” said Escartin.
But her optimism turned into fear after Sanofi admitted that persons who got injected with Dengvaxia and then contracted dengue for the first time might suffer from a severe form of the disease.
Escartin’s child received two shots of Dengvaxia before the Food and Drug Administration of the Philippines ordered Sanofi last December 5 to stop the sale and distribution of the vaccine.
But the government’s action isn’t allaying the fears of Escartin and her fellow Hagonoy residents. They said the vaccine had worsened their anxiety. Before, they only had to worry about dengue. But now, besides the illness, they also dread the effects of the supposed prevention against dengue.
WATCH NEWS5’S VIDEO REPORT:
Senate probe of Dengvaxia mess
At the Senate, lawmakers are getting ready to grill those who are being linked to the Dengvaxia mess.
Senator Joseph Victor “JV” Ejercito on Thursday said he wanted to focus on the possibility of “haste” in the drug’s purchase after he was asked by reporters if former President Benigno Aquino III should speak up on the issue.
The Philippines, during the Aquino administration, was the first Asian country that approved the commercial sale of the vaccine in December 2015.
In January 2016, then Department of Health (DOH) Secretary Janette Garin announced that Aquino had approved the administration of P3.5 billion worth of Dengvaxia to 1,077,623 nine-year-old public school students in Metro Manila, Central Luzon, and Calabarzon.
The vaccine arrived in the country in February 2016, two months before the DOH launched the school-based dengue vaccination program in April.
“Sa ngayon kasi [For now], we want to focus on the procurement, kung nagkaroon ng haste sa [if there was haste in the] procurement. It will be premature to point to the top already,” said Ejercito.
“But if it will reach the top then we will advise them to answer,” he added.
Ejercito said Aquino’s meetings with Sanofi Pasteur executives in France and China is something that should be looked into.
“Bakit there were two instances, hindi lang sa France, ‘yong isa sa China [Why were there two instances, not only in France but also in China]? What (were) the officials of Sanofi doing in China while the President (was) there?” he asked.
In 2014, Aquino met with Sanofi officials in China, who informed him that they were developing vaccines against dengue. In 2015, he also met with Sanofi Pasteur CEO Olivier Charmeil for a courtesy call at the Hotel Scribe in Paris, France.
The Senate is scheduled to conduct a public hearing on the Dengvaxia controversy on December 11.
Ejercito said it would be up to the Senate Blue Ribbon to decide on whether it would ask Aquino to attend the inquiry.
“Call na po siguro ‘yan ng Blue Ribbon because it will fall on their accountability,” he said.
WATCH NEWS5’S VIDEO REPORT: