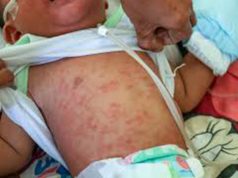
In the wake of various issues surrounding the dengue vaccine Dengvaxia, the World Health Organization (WHO) has released an updated posting of Frequently Answered Questions to lend clarity regarding the clinical trials, regulatory approvals, and efficacy of the dengue vaccine.
The full FAQ can be found here.
In this updated Questions and Answers (Q&A), WHO acknowledges that the dengue vaccine can have significant population-level benefits in highly endemic areas and recommended continued vaccination of individuals who have started vaccinations.
“In documented high seroprevalence settings, where vaccination has started but the schedule has not yet been completed, there is likely to be an overall benefit to the population if individuals complete the schedule, hereby assuring protection of seropositive individuals who make up the majority of the vaccinated population,” the WHO guidance said.
Given that more than 85% of the Philippine population has had previous exposure to dengue, the guidelines confirm that the dengue vaccine accords protection to the majority of Filipinos.
WHO also urged authorities to work on developing a rapid diagnostic assay to determine past dengue infection. Although there are various tests available to determine serostatus, such as the igG ELISA test, these are complex to use and are not yet suitable for routine practice in the context of a public vaccination program.
The WHO guidelines pointed out that unvaccinated individuals who have previously been exposed to dengue and vaccinated individuals with no dengue exposure have a similar risk of developing severe dengue.
“There is no evidence that clinical manifestations of disease were more severe in vaccinated seronegative persons compared to unvaccinated seropositive persons. For the entire vaccinated population, overall, the risk of severe dengue is reduced compared to a non-vaccinated population,” it stated.
WHO noted the “legitimacy and adherence to regulations and parameters of the Phase 1-3 clinical trials to license Dengvaxia, and subsequent/continuous tests as standard practice for any vaccine.”
WHO has started engaging independent external experts to review the new data generated by Sanofi Pasteur in order to provide advice on revisions to the WHO policy position paper from 2016.
On 6-7 December 2017, the WHO Global Advisory Committee on Vaccine Safety (GACVS) reviewed the data and subsequently published a statement related to the safety of the product.
The review will lead to revised recommendations from the Strategic Advisory Group of Experts on Immunization (SAGE) in April 2018 on the use of the Dengvaxia vaccine. It will also lead to an updated WHO position paper on the dengue vaccine thereafter.
The WHO Secretariat recommends that the revised Q & A document be read in conjunction with its position paper on the dengue vaccine released in July 2016, which conditionally recommended the use of the dengue vaccine in areas where dengue is highly endemic. This is defined by seroprevalence, which refers to the proportion of people in a population who have already been infected. WHO developed the guidelines on how to determine seroprevalence in an area to help countries that were considering use of the vaccine.
For its part, Sanofi Pasteur has proposed a label change to the national regulatory authorities in the countries where Dengvaxia® has been licensed. The final wording of the amended label will be decided by the national regulatory authorities of the respective countries, including the Philippines.
Trial participants who were previously exposed to dengue at the time of the vaccination had “a durable protection against severe dengue and hospitalization during the entire 5-year observation period.”
Any further guidance, including a review by SAGE and update of the WHO position paper on Dengvaxia®, will likely be available no earlier than April 2018 after a rigorous review of the new data and additional activities, such as population based modeling, are undertaken.
To reduce risk of dengue, the WHO recommends that all individuals, regardless whether they have been vaccinated or not, should take personal protective measures to avoid mosquito bites and for those with clinical symptoms compatible with dengue virus infection, regardless whether they have been vaccinated or not, to seek prompt medical care.
Click and watch, below, a related report on dengue deaths from News5’s Mon Gualvez: